 What is MRD?
What is MRD?
Measurable residual disease (MRD) refers to the presence of leukemic cells below the threshold of detection when using conventional morphologic methods.1,2 While there is no consensus on a precise definition of MRD positivity, a sensitivity threshold of 10–4 has been used to predict patient outcomes.3
Complete remission (CR) is typically defined as < 5% bone marrow blasts.3 A substantial proportion of adult patients with ALL relapse despite achieving CR, with a reported relapse rate of 40%–50%.4 Furthermore, patients who achieve CR according to morphologic assessment alone may harbor some residual cancer cells in the bone marrow, with 30%–40% testing positive for MRD.5,6,*
MRD and risk of relapse7
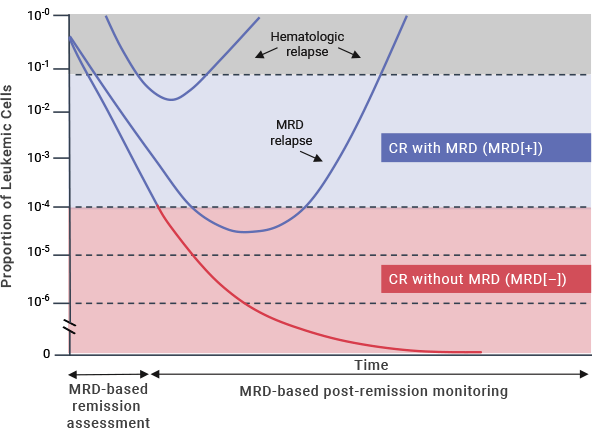

Research continues to lead to an improved understanding of the impact of MRD on patient prognosis and outcomes.8
 Why is MRD
Why is MRD
important?
A meta-analysis (Berry et al, 2017) assessed the presence of MRD after induction and consolidation therapy and concluded that MRD positivity was associated with negative outcomes regardless of treatment.8
According to the Berry meta-analysis, the presence of MRD is a strong predictor of relapse, which leads to limited treatment options and poor patient outcomes.8 Achieving MRD(–) status early in the course of treatment has been shown to give patients a reduced risk of relapse and a stronger chance for longer overall survival.8
Adult ALL and MRD
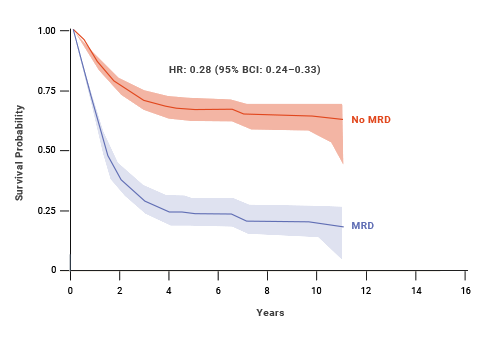
At 10 years, adult patients who achieved MRD negativity had 64% EFS (event-free survival) compared with 21% EFS for patients who were MRD(+).8,† An estimated benefit was also seen in OS with MRD negativity in adult patients.8
Impact of MRD status on EFS: adult ALL8,†
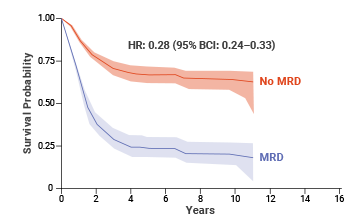
Impact of MRD status on OS: adult ALL8,‡
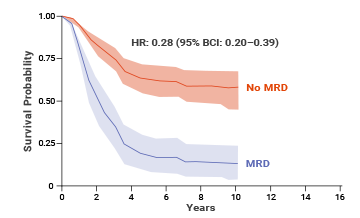
Pediatric ALL and MRD
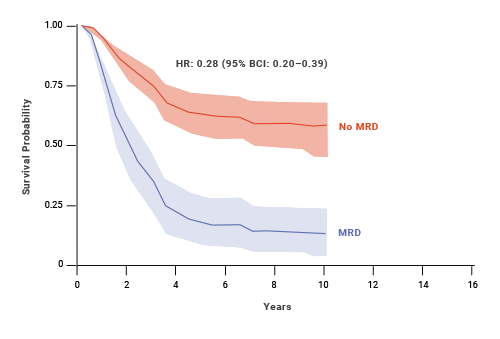
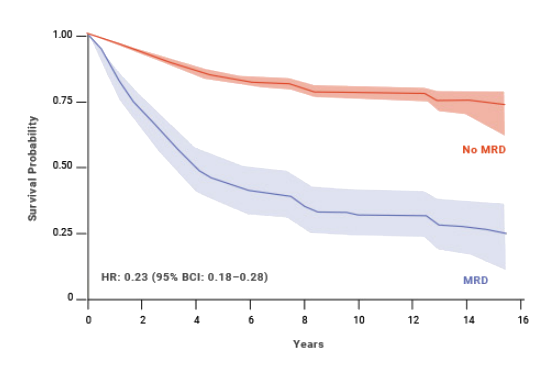
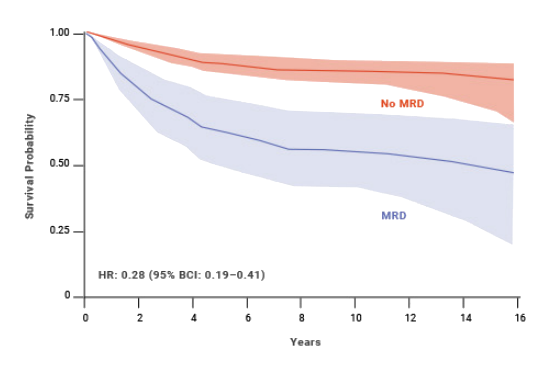
MRD status has also shown to be a predictor of OS and EFS in pediatric patients with ALL.8 At 10 years, pediatric patients who achieved MRD negativity had 77% EFS compared with 32% for patients who were MRD(+).8,§ An estimated benefit was also seen in OS with MRD negativity in pediatric patients.8
Impact of MRD status on EFS: pediatric ALL8,§

Impact of MRD status on OS: pediatric ALL8,**

MRD status is a prognostic factor for post-HSCT outcomes
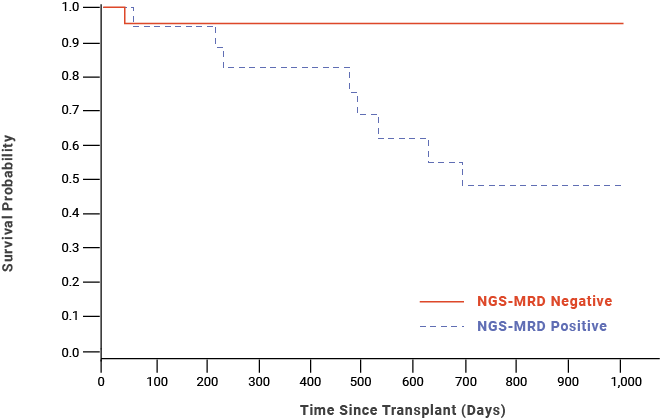
A study (Pulsipher et al, 2015) found that MRD negativity prior to HSCT was associated with improved 2-year OS probability in pediatric/AYA patients with B-cell precursor ALL.9,††
Post-HSCT OS probability for pediatric/AYA patients with B-cell precursor ALL9,††
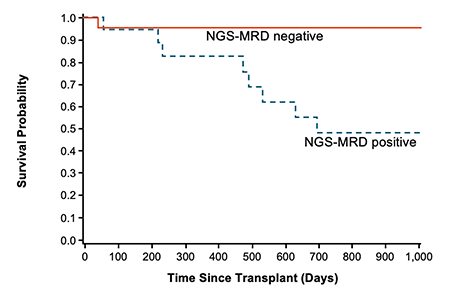
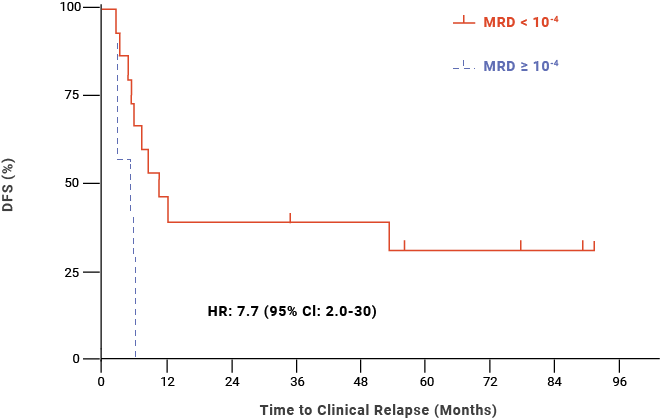
Additionally, a retrospective study (Logan et al, 2014) found that MRD ≥ 10-4 within 30 days prior to HSCT was significantly associated with relapse post-HSCT in adult/AYA patients with B-cell ALL.10,‡‡
Post-HSCT DFS probability for adult/AYA patients with B-cell ALL10,‡‡
Watch Jae Park discuss the
prognostic value of MRD


Associate Attending Physician
Memorial Sloan Kettering Cancer Center

